A decade after its discovery, the migrasome—a unique organelle enabling systematic, long-range, and targeted cellular communication—has begun to establish a new conceptual framework for cell–cell communication. This paradigm is reshaping our understanding of angiogenesis, coagulation, tumor metastasis, and immunity, while opening unprecedented avenues for diagnostics and therapeutic intervention.
The Missing Theory in Cell-Cell Communication
For decades, scientists have recognized two principal modes of intercellular communication: direct cell–cell contact and the secretion of soluble signaling molecules. The latter is largely explained by gradient theory, in which signaling molecules diffuse to form concentration gradients that guide cellular responses. Yet a fundamental question has persisted: soluble signals are inherently unstable—particularly in dynamic flow environments such as the circulation—so how do cells, especially those in distant regions of a complex organism, exchange information that is both stable and precisely controlled in space and time?
In 2014, a discovery at Tsinghua University provided an answer. While examining migrating cells, Li Yu and colleagues observed long membrane tubules bearing vesicle-like structures at their tips and intersections. Under electron microscopy, these structures resembled opened pomegranates. Because their formation depended on cell migration, they were named migrasomes.
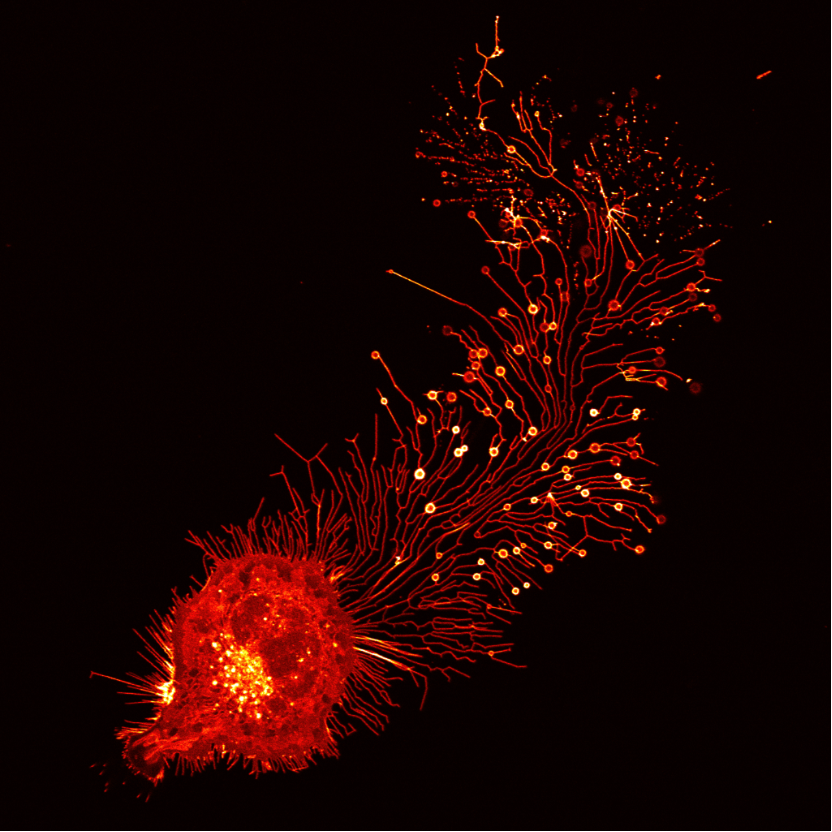
A confocal image revealed that a fibroblast generates migrasomes. Credit: Tsinghua University
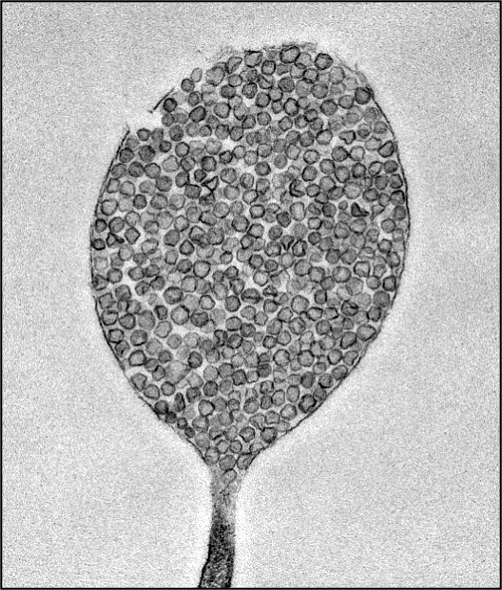
Transmission electron microscopy image reveals that migrasomes are packed with numerous small vesicles. Credit: Cell Research
Over the past decade, research has revealed that migrasomes function as signaling platforms left behind by migrating cells. A defined combination of signaling molecules is selectively transported into migrasomes and released in a highly controlled manner, enabling the delivery of combinatorial signals with spatial and temporal precision. In this way, migrasomes provide a long-sought mechanism for cells to dispatch packaged, location-specific information to distant or neighboring targets—bridging the gap between direct contact and diffusive secretion, and resolving a longstanding puzzle in cellular communication.
A New Theory of System-Level Targeted Communication
Migrasomes are far more than just another class of extracellular vesicle. These single-membrane organelles, typically 0.5–3 μm in diameter, carry a rich and selective cargo—chemokines, cytokines, growth factors, and other signaling molecules—while their surface can recruit factors such as components of the coagulation cascade, thereby directly influencing local blood clotting.
What distinguishes migrasomes is their functional versatility. While still tethered to retraction fibers, they act as spatially defined secretion platforms, releasing signals locally to generate linear gradients that guide neighboring cells. Upon detachment, they enter the extracellular milieu or circulation and serve as long-range delivery vehicles, transporting their molecular cargo to sites of injury or inflammation with remarkable precision.
This dual functionality—combining localized secretion with systemic transport—establishes a previously unrecognized, system-level communication network. In doing so, migrasomes offer a new conceptual framework for how cells achieve long-distance, high-precision information transfer beyond the limits of classical diffusion-based signaling.
Researchers have begun to uncover the elegant molecular machinery behind migrasome formation. The process happens in three stages: first, nucleation, triggered by the assembly of SMS2 spots; second, maturation, controlled by the PIP5K1α-Rab35-integrin axis; and third, expansion, driven by the formation of tetraspanin-rich microdomains. The coordinated action of these three steps ensures that migrasomes are built at the right time and place — laying the structural foundation for system-level targeted communication.
Beyond secreted proteins, the cargo carried by migrasomes is remarkably diverse, encompassing mRNAs, growth factors, coagulation factors, entire secretory vesicles, and even damaged mitochondria. Notably, cargo selection appears to be tightly regulated, underscoring migrasomes as highly versatile organelles. This breadth of cargo underlies their central role in cell physiology and highlights their potential importance in health and disease.
A wide range of functions has emerged from recent studies. In zebrafish embryos, migrasomes act as chemotactic cues that guide dorsal forerunner cells to form Kupffer’s vesicle, a structure essential for establishing left–right asymmetry. In developing chicken embryos, they promote angiogenesis, while in adult tissues, they contribute to vascular stability. Migrasomes can also initiate rapid clotting at sites of injury by presenting and releasing coagulation factors, enabling precise, spatiotemporally controlled hemostasis. In another line of work, damaged mitochondria are selectively packaged into migrasomes and expelled from migrating cells, representing a previously unrecognized mechanism of mitochondrial quality control.
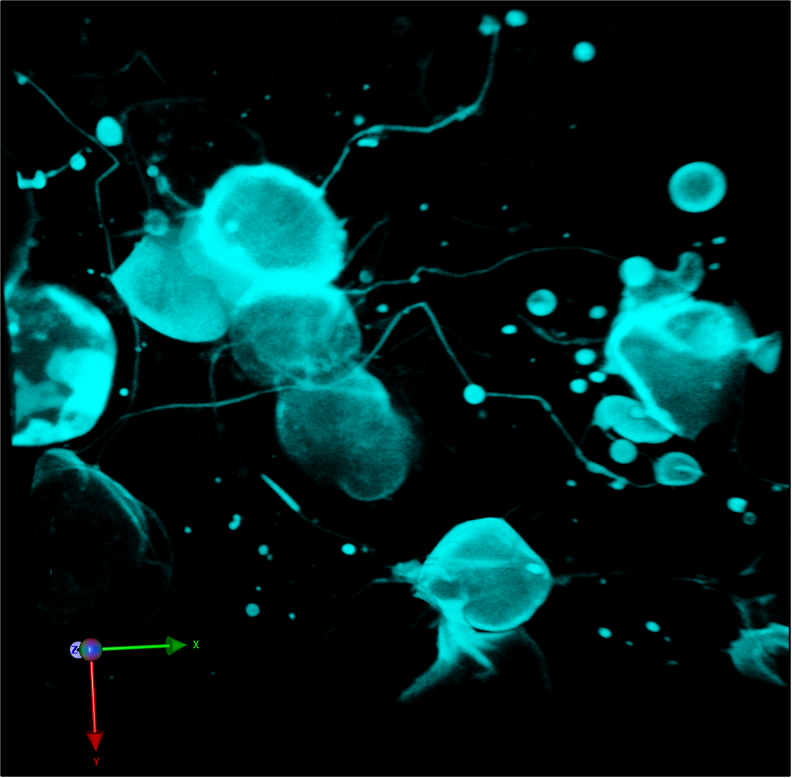
A three-dimensional confocal image shows that zebrafish embryonic cells generate migrasomes. Credit: Nature Cell Biology
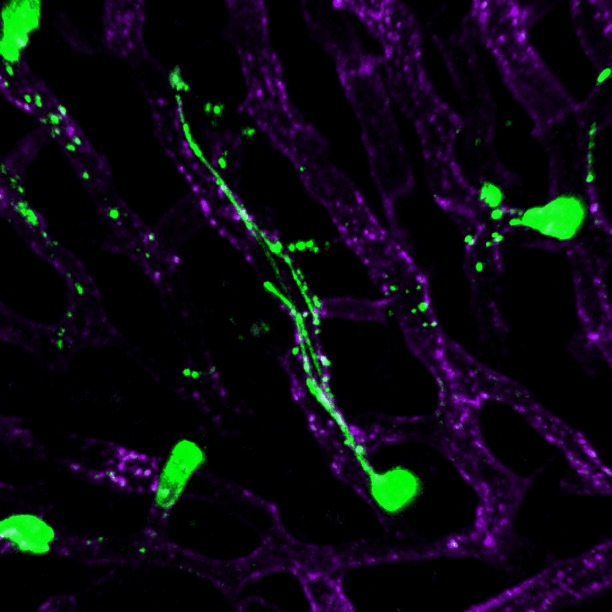
Intravital imaging of mice shows that neutrophils generate migrasomes within blood vessels. Credit: Nature Cell Biology
Over the past decade, research has revealed that migrasomes function as signaling platforms left behind by migrating cells. A defined combination of signaling molecules is selectively transported into migrasomes and released in a highly controlled manner, enabling the delivery of combinatorial signals with spatial and temporal precision. In this way, migrasomes provide a long-sought mechanism for cells to dispatch packaged, location-specific information to distant or neighboring targets—bridging the gap between direct contact and diffusive secretion, and resolving a longstanding puzzle in cellular communication.
A New Theory of System-Level Targeted Communication
Migrasomes are far more than just another class of extracellular vesicle. These single-membrane organelles, typically 0.5–3 μm in diameter, carry a rich and selective cargo—chemokines, cytokines, growth factors, and other signaling molecules—while their surface can recruit factors such as components of the coagulation cascade, thereby directly influencing local blood clotting.
What distinguishes migrasomes is their functional versatility. While still tethered to retraction fibers, they act as spatially defined secretion platforms, releasing signals locally to generate linear gradients that guide neighboring cells. Upon detachment, they enter the extracellular milieu or circulation and serve as long-range delivery vehicles, transporting their molecular cargo to sites of injury or inflammation with remarkable precision.
This dual functionality—combining localized secretion with systemic transport—establishes a previously unrecognized, system-level communication network. In doing so, migrasomes offer a new conceptual framework for how cells achieve long-distance, high-precision information transfer beyond the limits of classical diffusion-based signaling.
Researchers have begun to uncover the elegant molecular machinery behind migrasome formation. The process happens in three stages: first, nucleation, triggered by the assembly of SMS2 spots; second, maturation, controlled by the PIP5K1α-Rab35-integrin axis; and third, expansion, driven by the formation of tetraspanin-rich microdomains. The coordinated action of these three steps ensures that migrasomes are built at the right time and place — laying the structural foundation for system-level targeted communication.
Beyond secreted proteins, the cargo carried by migrasomes is remarkably diverse, encompassing mRNAs, growth factors, coagulation factors, entire secretory vesicles, and even damaged mitochondria. Notably, cargo selection appears to be tightly regulated, underscoring migrasomes as highly versatile organelles. This breadth of cargo underlies their central role in cell physiology and highlights their potential importance in health and disease.
A wide range of functions has emerged from recent studies. In zebrafish embryos, migrasomes act as chemotactic cues that guide dorsal forerunner cells to form Kupffer’s vesicle, a structure essential for establishing left–right asymmetry. In developing chicken embryos, they promote angiogenesis, while in adult tissues, they contribute to vascular stability. Migrasomes can also initiate rapid clotting at sites of injury by presenting and releasing coagulation factors, enabling precise, spatiotemporally controlled hemostasis (5). In another line of work, damaged mitochondria are selectively packaged into migrasomes and expelled from migrating cells, representing a previously unrecognized mechanism of mitochondrial quality control.
Read the full papers:
1. Cell Research. DOI: 10.1038/cr.2014.135
2. Cell Research. DOI: 10.1038/s41422-024-00992-7
3. Cell Discovery. DOI: 10.1038/s41421-024-00749-x
4. Nature Cell Biology. DOI: 10.1038/s41556-022-01026-3
5. Nature Cell Biology. DOI: 10.1038/s41556-024-01440-9
6. Nature Cell Biology. DOI: 10.1038/s41556-019-0358-6
7. Cell Research. DOI: 10.1038/s41422-023-00811-5
8. Nature Cell Biology. DOI: 10.1038/s41556-019-0367-5
9. Cell. DOI: 10.1016/j.cell.2021.04.027