Crossing the Boundary of Natural Protein Architecture
Being able to create any protein that can exist structurally doesn’t just help us lay the groundwork for understanding life’s basic rules—it also drives game-changing innovations in the next generation of medicines and protein-based materials. A chemical method using peptide hydrazide as the key “building block” has become an effective, widely used tool in modern de novo chemical protein synthesis—the process of making proteins from scratch using chemical reactions.
The Rising Need for “Beyond-Biology” Protein Synthesis
As the fundamental molecules that support all life on Earth, proteins are closely tied to human progress in biomedicine and advanced materials. While AI-driven de novo protein design has ushered in a revolutionary era for studying natural proteins, our next big challenge is to design and develop functional proteins that break free from the structural limits of biological systems. To do that, we need a key technique: de novo chemical protein synthesis. For a long time, though, this technique was only possible for a small group of experts—mostly because there was no easy-to-use building block for making proteins chemically.
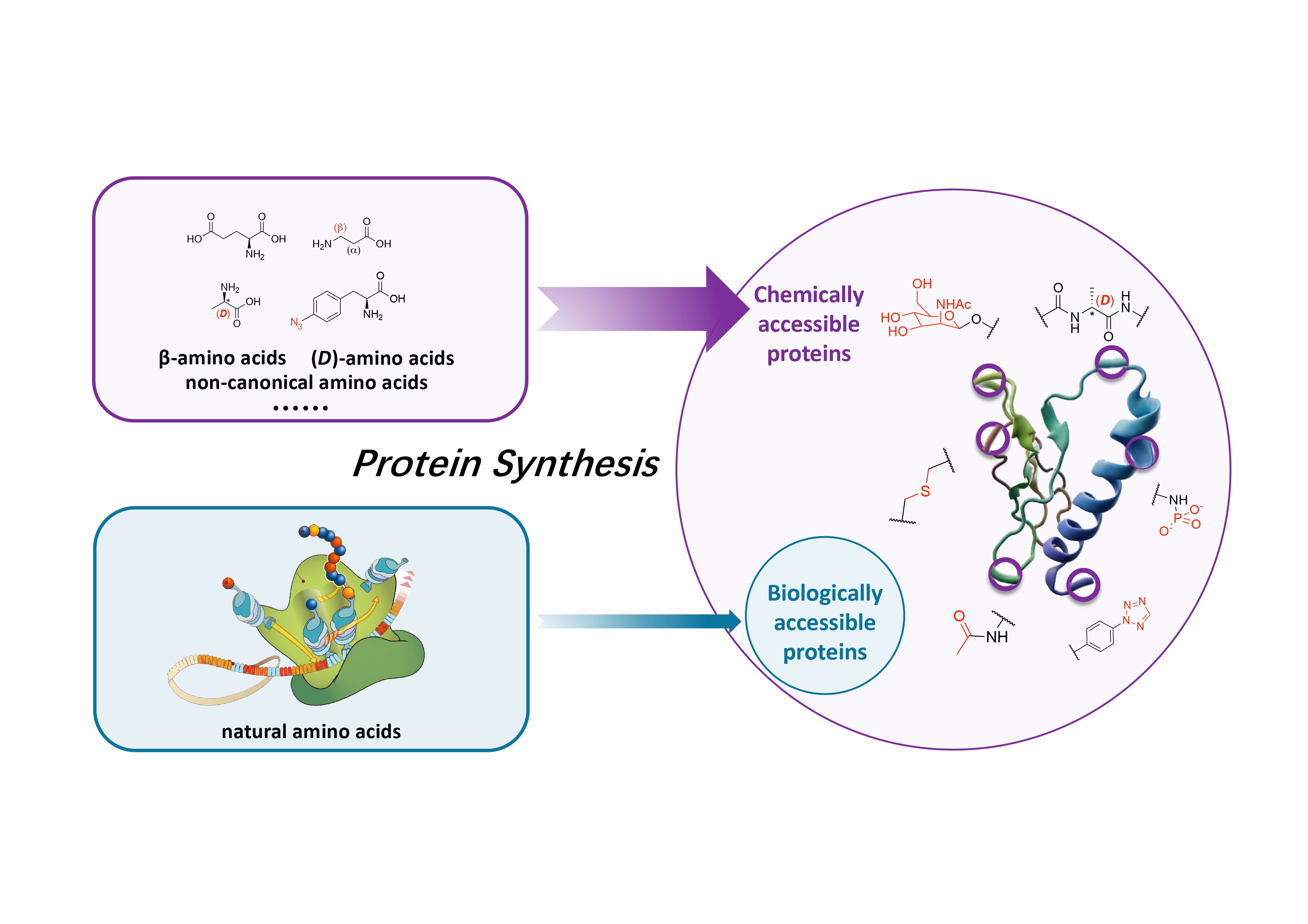
Chemical protein synthesis versus biological protein synthesis. Credit: Tsinghua University
Peptide Hydrazide Ligation: A Robust Core Technology
To build a comprehensive, widely accessible platform for de novo chemical protein synthesis, researchers from Tsinghua University invented a method called peptide hydrazide ligation. Here’s how it works in simple terms: a fully unprotected peptide hydrazide connects specifically with another matching peptide to form a longer peptide chain. By repeating this step over and over, scientists can build an entire, full-length protein. The biggest upside of this method is that peptide hydrazide is cheap and easy to obtain—it can be made in small amounts (just a few milligrams) or large batches (up to kilograms)—and it’s simple to handle. This makes chemical protein synthesis reliable, practical, and easy to use for more people.
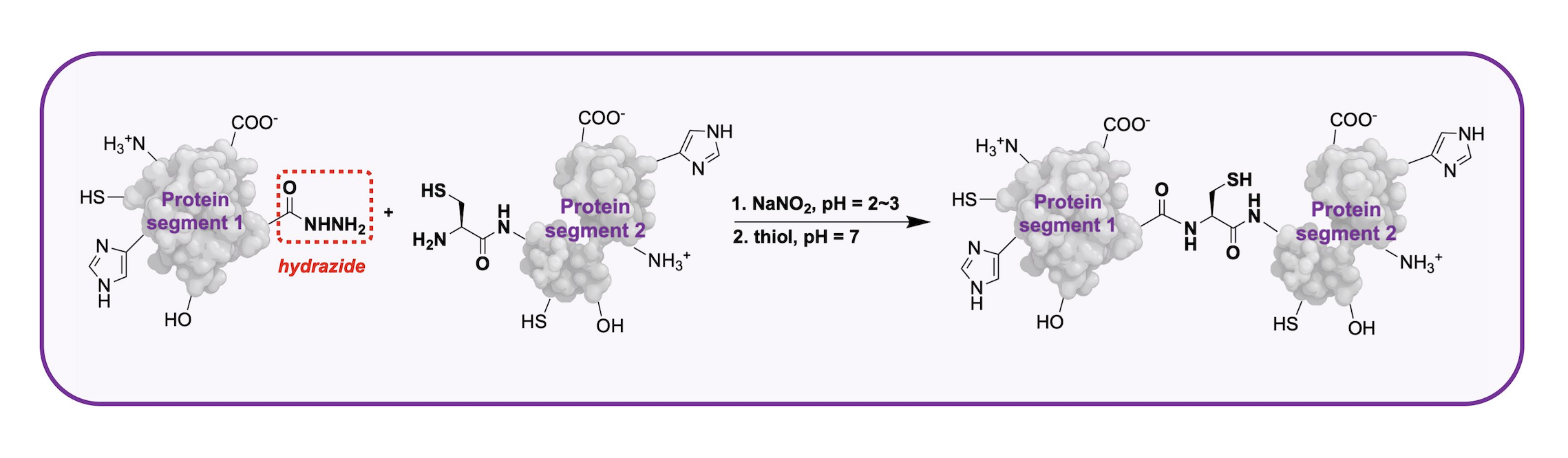
Peptide hydrazide ligation. Credit: Tsinghua University
Building on peptide hydrazide ligation, Tsinghua researchers developed a complete, powerful “toolbox” for de novo chemical protein synthesis, eventually creating a full technological platform. This innovative breakthrough has completely expanded what’s possible with total chemical protein synthesis: today, it can make proteins with around 900 amino acids, covering nearly 95% of all known proteins. Most importantly, it has made chemical protein synthesis accessible to more researchers—turning it from a specialized skill into a widely used technology.
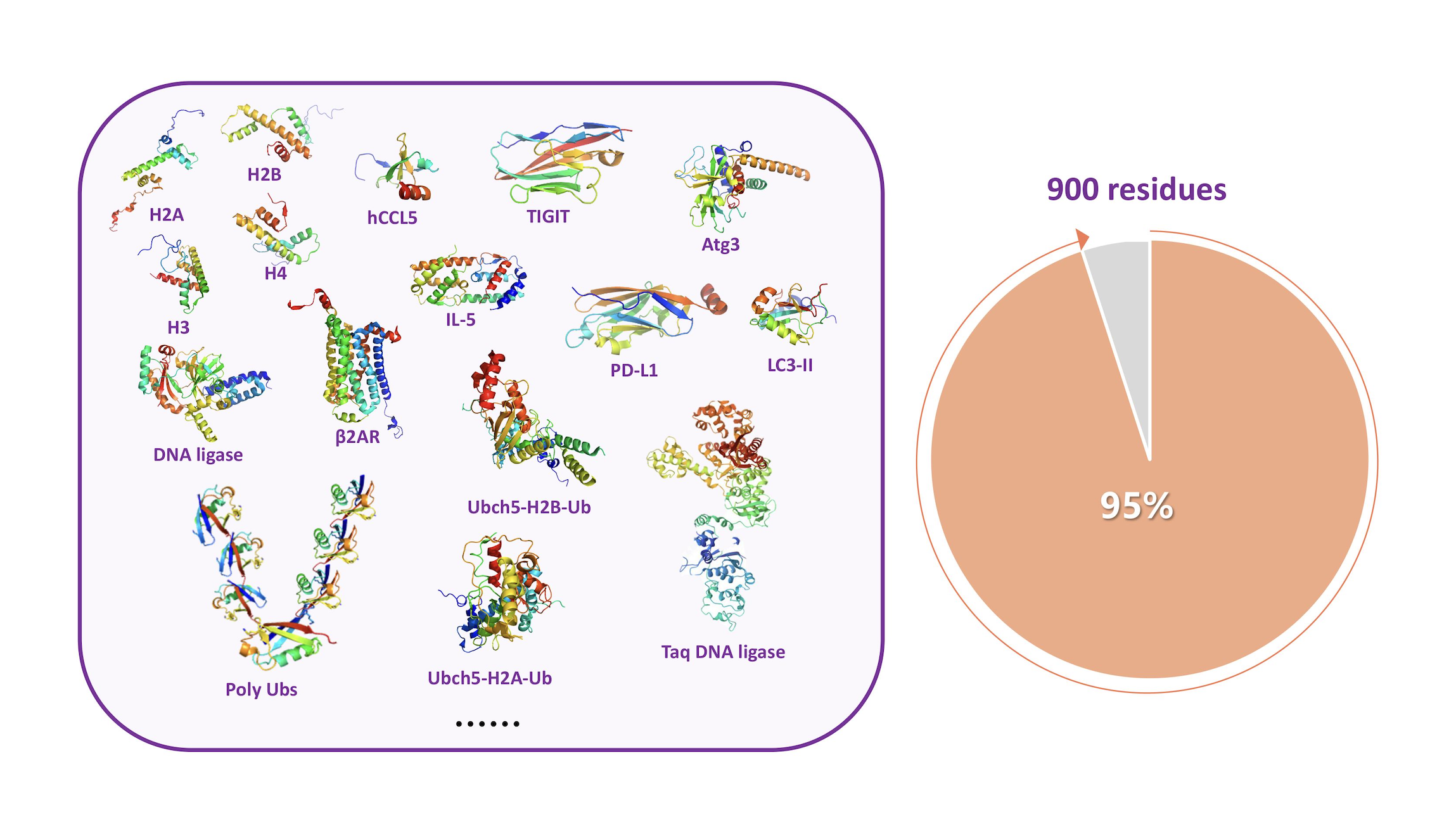
de novo chemical synthesis of proteins up to 900 amino acids. Credit: Tsinghua University
Increasing Applications of Hydrazide-Driven Chemical Protein Synthesis
Using peptide hydrazide ligation, Tsinghua researchers have shown its value in many areas: studying how proteins are modified after they’re made (known as post-translational modifications), developing biocatalysts, and creating therapeutic drugs. Today, peptide hydrazide ligation has become a go-to method for chemical protein synthesis, helping it spread from specialized expert labs to research and industrial fields all over the world.
Looking ahead, combining hydrazide chemistry with automated synthesis robots will make de novo chemical protein synthesis faster and more efficient. Artificial intelligence (AI) will use data on the structure and properties of chemically synthesized proteins—including those with non-natural parts—to help design peptide or protein drugs and materials that work even better than natural ones. These chemically made “beyond-biology” proteins are opening a new chapter in how we use proteins to improve human health and well-being.
Read the full papers:
1. Angewandte Chemie International Edition, DOI: 10.1002/anie.201100996
2. ACS Central Science, DOI: 10.1021/acscentsci.4c00642