A new mesoscale imaging paradigm integrates digital adaptive optics, scanning light-field microscopy, and deep learning to capture 3D cellular behaviors and functions across whole organs with unprecedented fidelity.
For decades, biological imaging has been defined by a fundamental struggle between two axes: resolution and field of view. While breakthroughs like cryo-EM and super-resolution microscopy have pushed the boundaries of biological imaging to see individual molecules, they are typically limited to a few cells. Conversely, medical imaging such as CT and MRI can visualize entire organs, yet they lack the single-cell resolution necessary to understand complex cellular interactions.

Tradeoff between resolution and field of view for biomedical imaging. Credit: Tsinghua University
It represents a significant gap in the technical landscape. Capturing both a large field of view and high resolution to analyze large-scale intercellular interactions at the organ level is known as mesoscale imaging. Bridging this gap is essential for understanding how large-scale intercellular dynamics govern life. Now, a research team at Tsinghua University has developed RUSH3D, a next-generation mesoscale intravital fluorescence microscopy.
A breakthrough in mesoscale computational imaging
At the core of RUSH3D is a transformative approach to digital adaptive optics (DAO), which overcomes the fundamental challenge of spatially nonuniform optical aberrations. While traditional microscopes sacrifice resolution as the field of view expands, the team implemented a wave-optics DAO framework capable of performing tiled, high-speed aberration corrections across the entire imaging volume. This innovation allows the system to maintain near-diffraction-limit resolution even at a centimeter-wide mesoscale, an essential requirement for capturing fine cellular details throughout whole organs. The aberration correction capability is seamlessly integrated into a scanning light-field imaging framework, further enhanced by axially elongated line-confocal illumination to suppress background fluorescence. By removing complex aberrations digitally rather than through bulky hardware, the RUSH3D provides the unprecedented fidelity for long-term, high-speed 3D observation in native intravital environments.
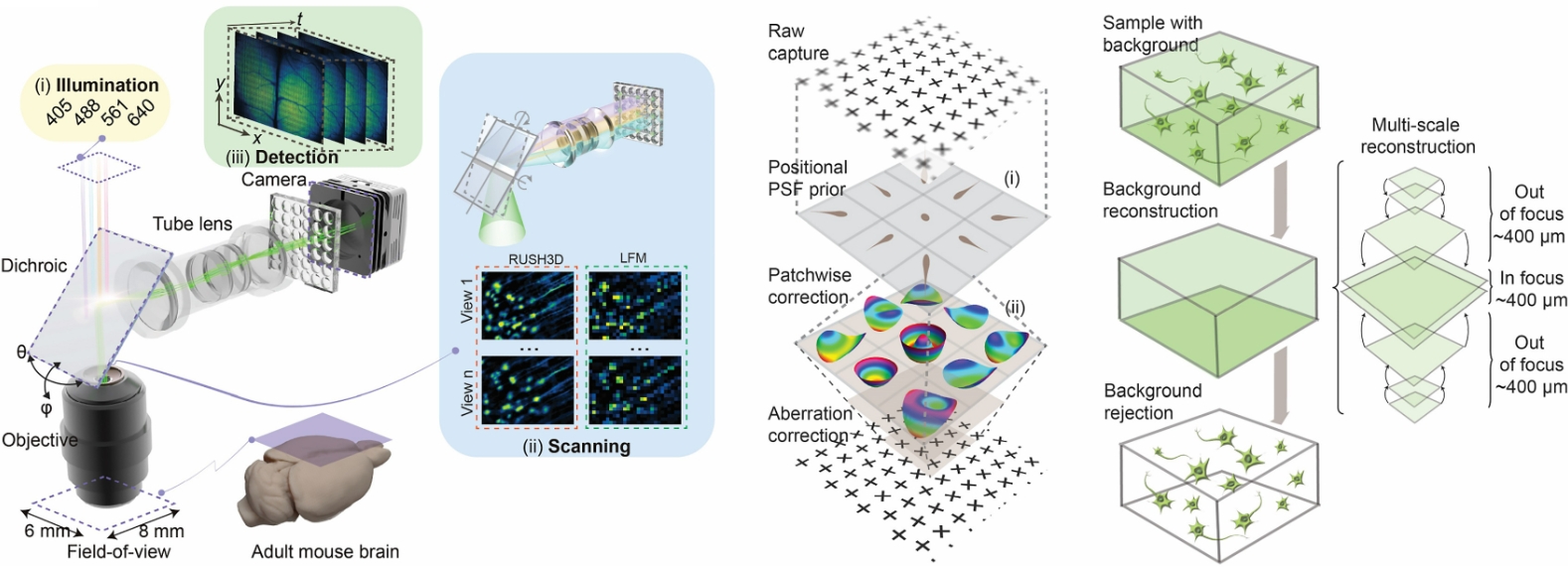
The RUSH3D framework. An illustration of the system architecture (left) highlighting the wave-optics DAO (right) for large-scale aberration removal, enabling high-fidelity 3D imaging across mesoscale biological volumes. Credit: Tsinghua University
AI-powered image processing and analysis
To handle the vast volumes of data generated at the mesoscale, RUSH3D leverages cutting-edge deep learning algorithms. The team implemented DeepCAD-RT for real-time noise suppression, enabling high-sensitivity imaging even under low light to minimize phototoxicity. Furthermore, the DeepWonder framework supports the rapid, automated extraction of cellular signals from thousands of cells simultaneously. The integrated pipeline ensures that mesoscale big data is translated into clear biological insights with minimal manual influence.
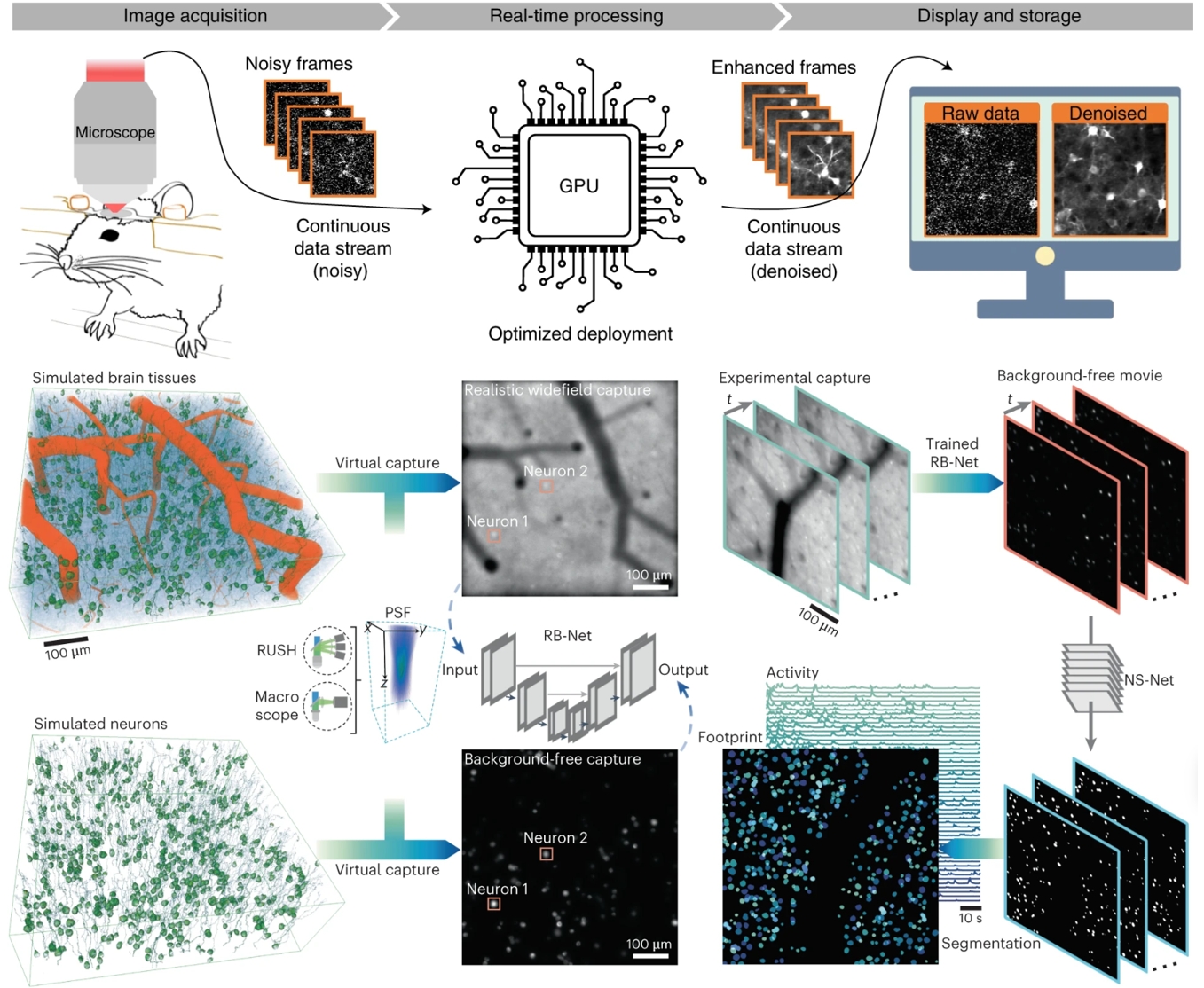
AI-powered image processing pipeline for denoising (top) and neural activity extraction (bottom). Credit: Tsinghua University
Visualizing cellular dynamics within the living organs
By integrating these novel technologies, RUSH3D allows for long-term, high-speed 3D imaging at a centimeter-scale field of view, which is a “game-changer” for observing intercellular dynamics across a mammalian organ.
“Modern AI and computational imaging models can fundamentally change the way we observe the complexity of life,” says the research team. “By combining hardware innovation with deep learning, we can now monitor biological processes that were previously invisible.”
The team has successfully recorded the activity of over 10,000 neurons simultaneously across multiple cortical areas in mice. Furthermore, they observed the formation and progression of multiple germinal centers in lymph nodes, capturing the real-time interactions between T cells and B cells during an immune response over several days.
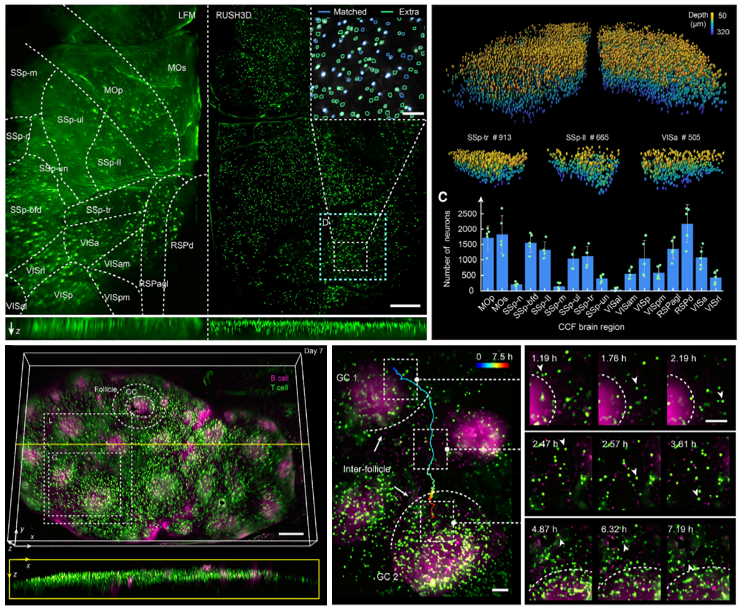
Mesoscale Intravital observations. (Top) Cortex-wide 3D neural recording in a behaving mouse. (Bottom) Long-term tracking of intercellular dynamics and B cell clustering within lymph node germinal centers. Credit: Tsinghua University
Future frontiers for advancing drug R&D
The development of RUSH3D marks a pivotal shift in how we observe the complexities of life at the mesoscale, opening doors to previously unreachable research frontiers. By capturing intricate interactions in native physiological environments, RUSH3D will offer researchers unprecedented visual evidence to evaluate therapeutic efficacy and safety during drug R&D, moving beyond barriers to offer a much clearer window into the performance of next-generation therapeutic candidates.
Read the full paper:
Cell, DOI: 10.1016/j.cell.2024.08.026