A fragmented understanding of how mosquitoes locate infected hosts and support viral transmission has limited effective control strategies. Integrated insights into host-derived signals, extracellular vesicle–mediated infection, and symbiont-based interference now provide a mechanistic framework for system-level intervention.
Mosquito-borne diseases such as dengue continue to impose a substantial global health burden, infecting hundreds of millions of people each year. At the heart of these diseases lies a deceptively simple but highly efficient biological cycle: a mosquito bites an infected person, acquires the virus, and later transmits it to a new host through another bite. This “host–mosquito–host” transmission cycle underpins the global spread of dengue and other arboviruses.
Mosquitoes play an active and highly selective role in determining whether a virus can spread. First, mosquitoes locate infected hosts within large populations. After ingesting infected blood, the virus replicates and disseminates within the mosquito before it can be transmitted again. Importantly, not all mosquitoes can transmit all viruses. For example, Aedes mosquitoes efficiently spread the dengue virus, whereas other species, such as Culex mosquitoes, cannot transmit the dengue virus. This species specificity has long been recognized; however, its underlying biological basis remains unclear.
These complexities point to two fundamental questions: how mosquitoes preferentially target infected individuals and what determines whether a mosquito becomes a competent vector? Addressing these questions is essential, as they represent the key bottlenecks that control transmission efficiency. Recent advances from three complementary lines of research now provide a more complete and mechanistic view of this process.
Dengue virus enhances host attractiveness to promote mosquito targeting
The first breakthrough reveals that the dengue virus actively enhances its own transmission at the host level. Infection alters the skin microbiota, leading to increased production of volatile compounds such as acetophenone, a microbiota-derived metabolite. This compound strongly stimulates mosquito olfactory systems, enabling mosquitoes to preferentially locate infected individuals—even when they are rare within a population. This finding explains a long-standing epidemiological puzzle: how mosquitoes efficiently identify infected hosts during the early stages of an outbreak. By biasing mosquito feeding behavior toward infectious individuals, the virus effectively increases its chances of being acquired and transmitted.
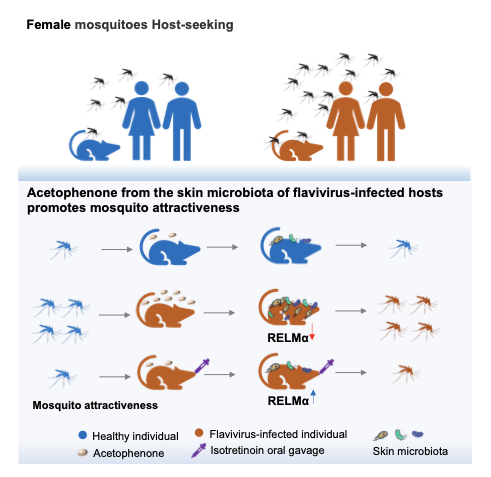
Dengue virus enhances host attractiveness by altering skin microbiota-derived volatiles, acetophenone, thereby promoting mosquito targeting of infected individuals. Designed by a team at Tsinghua University. Credit: Tsinghua University
Dengue virus establishes systemic infection in mosquitoes despite the acidic internal environment
The second advance addresses what happens after the mosquito takes an infectious blood meal. Inside the mosquito, the virus encounters a hostile environment: the hemolymph is acidic (pH ~6.0), a condition that can inactivate free viral particles. This challenges the traditional view that viruses spread as intact particles within the vector. Instead, the dengue virus uses a non-classical strategy. It hijacks extracellular vesicles (EVs) to transport its genetic material in a protected form. This process depends on a specific interaction between a mosquito protein, VCP, and the viral capsid. Notably, this interaction varies between mosquito species, providing a molecular explanation for why only certain mosquitoes can transmit the dengue virus. This work redefines how viruses survive and disseminate within the mosquito and reveals the molecular basis of vector specificity.
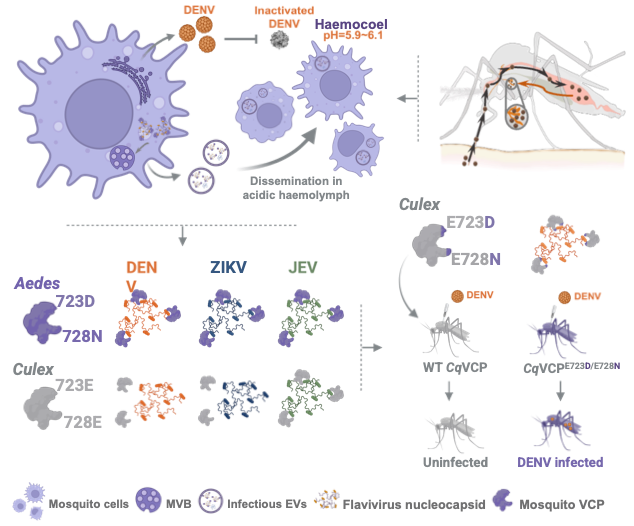
Dengue virus establishes systemic infection in mosquitoes through an extracellular vesicle–mediated mechanism that protects viral components from the acidic hemolymph environment. Designed by a team at Tsinghua University. Credit: Tsinghua University
Symbiont-based strategies effectively block dengue virus transmission
The third line of research translates these mechanistic insights into a practical intervention strategy. Researchers identified a naturally occurring symbiotic bacterium, Rosenbergiella sp. YN46, that colonizes the mosquito gut. This bacterium acidifies the gut environment during blood feeding, lowering the pH to levels that effectively inactivate the dengue virus before it can establish infection. Semi-field studies show that introducing this bacterium into mosquito breeding environments significantly reduces viral infection rates in mosquito populations. Because the bacterium is naturally occurring and environmentally stable, this approach represents a promising and scalable strategy for dengue control. As noted by the researchers, “Implementing an environmental intervention using a naturally occurring symbiotic bacterium represents an eco-friendly biocontrol strategy with promise for controlling dengue transmission in endemic regions.”
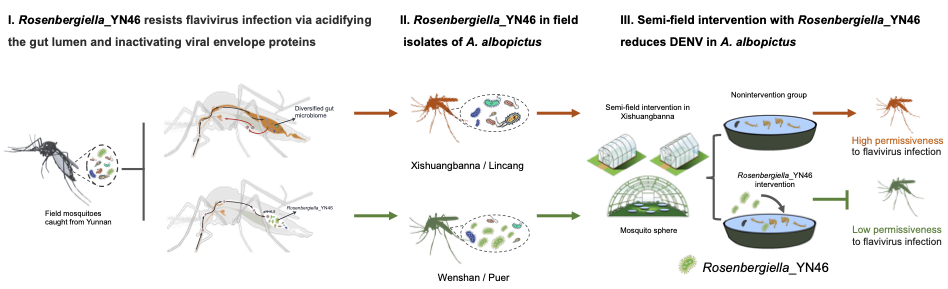
The naturally occurring symbiotic bacterium Rosenbergiella_YN46 inhibits dengue virus transmission by acidifying the mosquito gut during blood feeding. Designed by a team at Tsinghua University. Credit: Tsinghua University
Future directions
Together, these three lines of work establish a coherent, multi-scale framework for understanding dengue transmission: host-derived signals enhance mosquito attraction, EV-mediated mechanisms enable viral survival and dissemination within the mosquito, and symbiotic bacteria can disrupt infection at the earliest stage. This integrated perspective shifts the paradigm from isolated interventions to system-level control strategies. Future efforts should focus on integrating these strategies into coordinated intervention programs, evaluating their effectiveness across diverse ecological settings, and assessing long-term evolutionary responses. By targeting multiple rate-limiting checkpoints within the transmission cycle, this integrated framework provides a foundation for next-generation, system-level strategies to achieve sustainable control of dengue and other mosquito-borne viruses.
Read the full papers:
1. Cell, DOI: 10.1016/j.cell.2022.05.016
2. Nature, DOI: 10.1038/s41586-026-10100-x
3. Science, DOI: 10.1126/science.adn9524