The efficient and low-carbon recycling of spent lithium-ion batteries (LIBs) and plastic waste is a crucial challenge for achieving sustainable development in the new energy industry. Recently, Bing Deng’s research group from the School of Environment at Tsinghua University proposed a novel synergistic recovery method based on flash Joule heating (FJH)-enabled electrothermal chlorination. This method achieves high-selectivity separation and recovery of lithium and transition metals from spent LIBs within tens of seconds, demonstrates its universality for recycling different types of LIBs, and simultaneously converts the carbon resources in polyvinyl chloride (PVC) plastic into high-value graphene carbon materials.
With the rapid adoption of electric vehicles and portable electronics, lithium-ion batteries have become one of the core technologies supporting the energy transition. However, the concentrated emergence of large-scale end-of-life batteries poses severe environmental and resource challenges. Spent LIBs are rich in strategic critical metals such as lithium, cobalt, nickel, and manganese. These metals not only have highly concentrated resource endowments but their traditional extraction processes are accompanied by significant energy consumption and environmental burdens. Meanwhile, battery recycling often generates substantial plastic waste. Among these, PVC plastic, due to its chlorine content, is prone to producing corrosive and potentially harmful by-products during thermal treatment, making it a long-standing challenge in waste plastic management. Current mainstream battery recycling technologies primarily include pyrometallurgy and hydrometallurgy. Pyrometallurgy features a simple process but suffers from high energy consumption and carbon emissions, with light elements like lithium easily lost. Hydrometallurgy achieves higher recovery rates but relies heavily on acid and alkali reagents, involving complex processes and generating secondary wastewater. Concurrently, plastics are often merely consumed as fuel or reducing agents in most battery recycling systems, leaving the value of their carbon resources underutilized.
Addressing the above issues, Bing Deng's research group from the School of Environment at Tsinghua University proposed an Electrothermal Chlorination synergistic recovery strategy. This method treats spent LIB cathode materials and PVC plastic within the same electrothermal reaction system, achieving synergistic transformation and high-value utilization of multiple waste streams. Based on flash Joule heating technology, the method uses pulsed electric current to rapidly heat the reaction system to 400℃-2000 ℃ within an extremely short time, followed by rapid cooling. This allows precise control over the chlorination reaction process at both thermodynamic and kinetic levels. Figure 1 illustrates the overall principle and reaction pathway of the electrothermal chlorination synergistic recovery process. Under electrothermal conditions, PVC rapidly pyrolyzes, releasing chlorine sources like HCl and active reductive gases. This causes lithium in the LIB cathode materials to be selectively chlorinated into water-soluble lithium chloride, while transition metals such as cobalt, nickel, and manganese are reduced to their metallic or oxide forms. Leveraging differences in the thermodynamics of chlorination reactions and the solubility of products for different metals, the research team achieved efficient separation of lithium from transition metals: lithium can be extracted via simple water leaching, while transition metals can be recovered under mild acid leaching conditions, thereby realizing selective recovery of both.
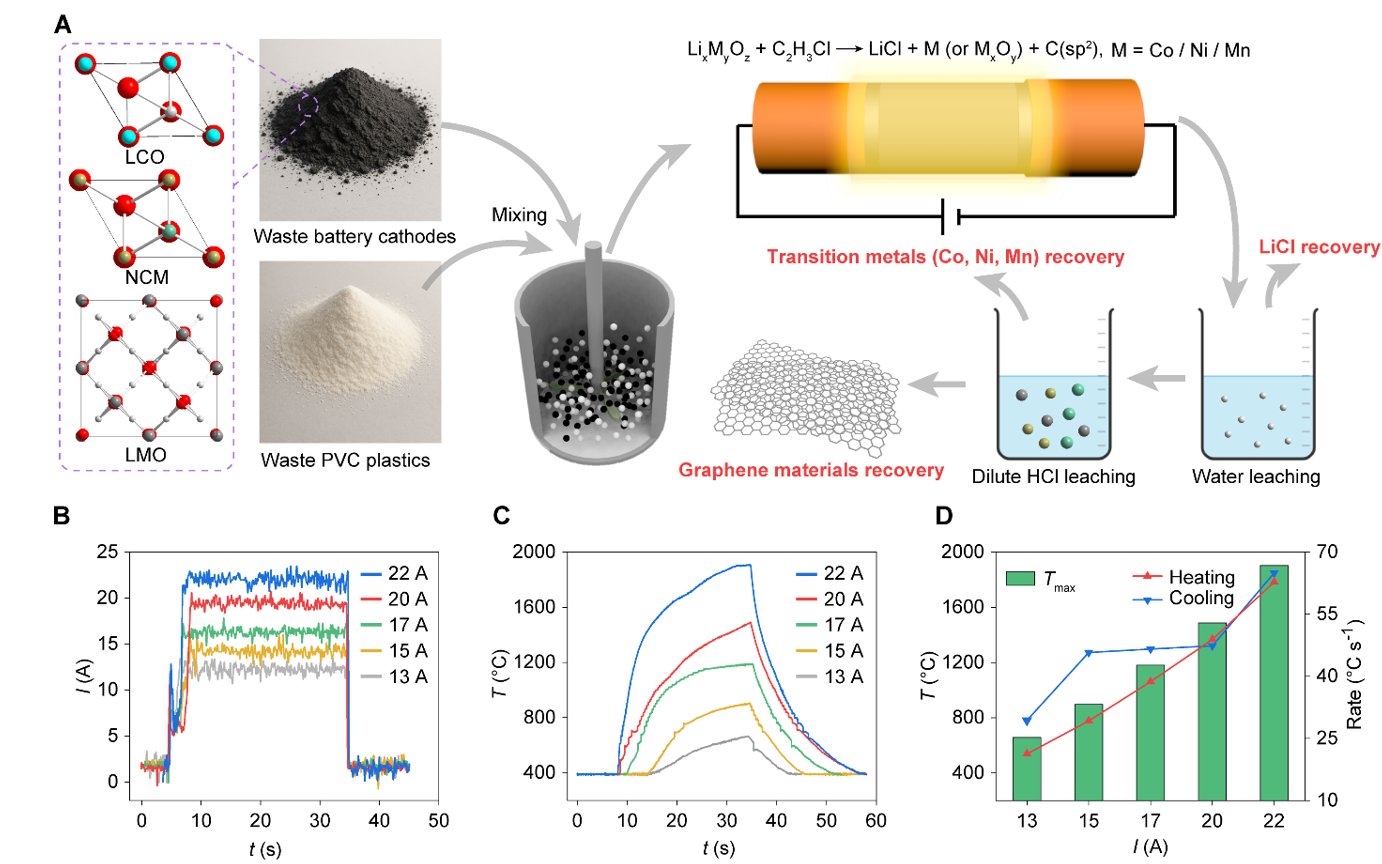
Figure 1. Synergistic recovery of spent lithium-ion batteries and PVC plastic based on electrothermal chlorination.
In experimental validation, the research team systematically investigated several mainstream cathode material systems, including LiCoO₂, LiMn₂O₄, and LiNixCoyMn1−x−yO₂. The results indicate that this method exhibits good applicability to different battery chemistries. Recovery rates for lithium and transition metals generally reached 94%–99%, with the lithium-transition metal separation coefficient reaching as high as ~2600, demonstrating extremely high selectivity and stability. Furthermore, this electrothermal chlorination process achieves high-value conversion of the carbon resources in PVC plastic alongside metal recovery. Under high-temperature electrothermal conditions and the catalytic effect of transition metals, the sp³ carbon structure in PVC is rearranged into a highly ordered sp² carbon structure, generating graphene materials with a layered structure. Structural characterization shows that the graphene material possesses good crystallinity and properties, offering a new "waste-to-material" pathway for plastic waste.
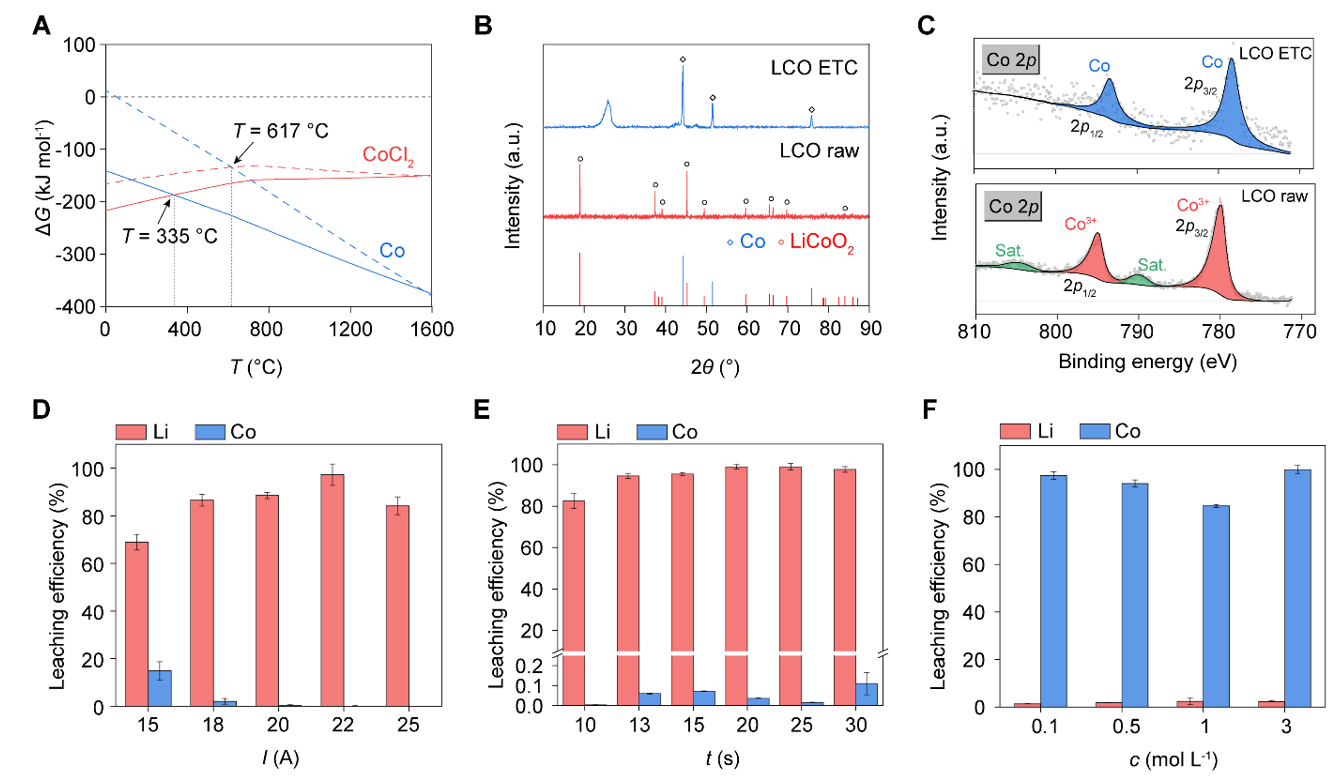
Figure 2. Electrothermal chlorination enables selective separation and recovery of lithium and transition metals.
The study also evaluated the engineering application potential through systematic techno-economic analysis and life cycle assessment. The results show that compared to traditional hydrometallurgical and pyrometallurgical recycling processes, the electrothermal chlorination synergistic recovery method has the potential to reduce carbon emissions by over 80%. Simultaneously, operational costs are significantly lowered due to reduced consumption of chemicals like acids and alkalis. Additionally, the research team has successfully scaled up reactor validation at the laboratory scale and constructed a kilogram-scale flash Joule heating reaction setup, laying the groundwork for subsequent pilot and engineering applications. This research provides a new technological paradigm for the synergistic treatment of spent lithium-ion batteries and chlorinated plastics, and offers a fresh perspective for the low-carbon, high-value circular utilization of multi-source solid waste.
On January 1, 2026, this research was published in the Cell Press journal Cell Reports Physical Science under the title “Co-recycling of waste lithium-ion batteries and polyvinyl chloride plastics.” Assistant Professor Bing Deng from the School of Environment is the corresponding author. Teng Wang, an undergraduate from Tanwei College, is the first author. Co-authors include Ph.D. candidates Erkang Feng, Zhenyu Ren, Mingyue Xu, and Yunxuan Zeng, research assistant Ziyu Huang, master’s student Hao Bi, and undergraduate Chun Huang from Xiuzhong College, all from the School of Environment.
The research was supported by the Tsinghua University-Toyota Joint Fund, the Beijing Natural Science Foundation, the National Natural Science Foundation of China, and others.
Link to paper: https://doi.org/10.1016/j.xcrp.2025.103048
Editor: Li Han

