The research group led by Prof. Wei Xie from the School of Life Sciences at Tsinghua University published a study on identifying a key transcription factor NR5A2 that bridges zygotic genome activation to the first lineage segregation during early embryonic development in the journal Cell Research on November 7, 2023. The study unveils critical roles for transcription factors in the transition from totipotency to pluripotency in early embryo development.
The journey of mammalian life begins with the fusion of sperm and eggs, culminating in the formation of a totipotent embryo through the process of fertilization. The first transcriptional event in the fertilized egg is referred to as zygotic genome activation (ZGA). Subsequently, the first lineage segregation occurs as development progresses, giving rise to inner cell mass (ICM) and trophectoderm (TE). However, how ZGA is connected to the first lineage segregation in mammalian embryos remains unclear.
NR5A2, a nuclear receptor family transcription factor, was strongly induced during mouse ZGA, with its DNA binding motif enriched in open chromatin during the 2-8 cell stages. In this study, researchers found embryos with Nr5a2 knockdown or knockout using base-editing arrested at the morula stage (16 cell). Transcriptome analysis revealed an essential role of NR5A2 in genes specifically activated during the 4-8 cell stage, but not those activated at the 2-cell stage during ZGA. Subsequently, researchers examined genome-wide binding of NR5A2 in mouse 2- and 8-cell embryos using CUT&RUN, and chromatin accessibility using ATAC-seq. Their findings included: 1) NR5A2 binds cis-regulatory elements and activates gene expression during the 2-8 cell stage; 2) NR5A2 binding peaks strongly enrich for B1 repetitive elements, which contain abundant transcription factor binding motifs for early embryonic development, such as OBOX, NR5A2, RARG; 3) NR5A2 has a minimal role in opening chromatin at the 2-cell stage, but is responsible for opening 8-cell specific regulatory elements. Many of these sites are bound by pluripotency factors such as NANOG, SOX2, and OCT4 in late-stage embryos or ESCs. Importantly, NR5A2 in the 8-cell stage regulates the expression of key pluripotency genes such as Nanog and Pou5f1/Oct4, as well as critical genes related to the regulation of the trophectoderm and primitive endoderm, including Tead4, Gata3, and Gata6. In summary, this study discovered NR5A2 as a key regulator in connecting ZGA and the first lineage segregation during embryonic development.
Prof. Wei Xie is the corresponding author of this paper. Ph.D students Fangnong Lai, Lijia Li, Xiaoyu Hu, and a postdoctoral fellow Bofeng Liu from the School of Life Sciences at Tsinghua University are co-first authors. Collaborators include Prof. Hongyu Deng and Huabin Tian from the Institute of Biophysics, Chinese Academy of Sciences, and Prof. Jinsong Li's and Qing Li from the Center for Excellence in Molecular Cell Science, Chinese Academy of Sciences. Ph.D student Ziqi Zhu from the School of Life Sciences at Tsinghua University and research associate Liu Ling from Prof. Wei Xie's research group also made important contributions to this study. This study also received help from the Animal Center and the Bioinformatics core facility at Tsinghua University. Dr. Bofeng Liu is a ShuiMu Scholar in Tsinghua University. Funding for this research was provided by the National Natural Science Foundation of China, the Key Research and Development Program of the Ministry of Science and Technology, and the Tsinghua-Beijing Life Science Center. Prof. Wei Xie is an HHMI International Research Scholar and a New Cornerstone investigator.
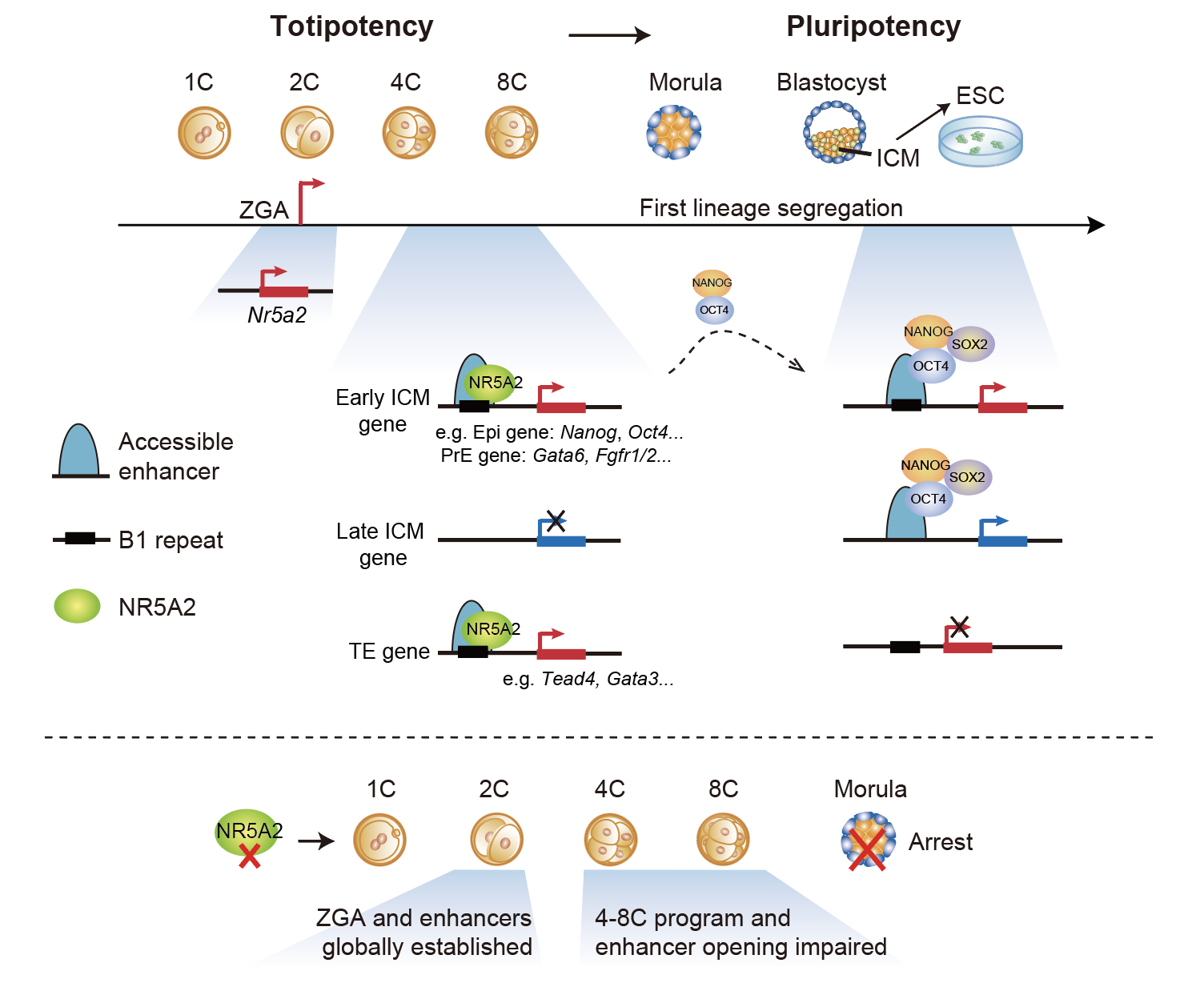
Fig.1 NR5A2 connects ZGA to the first lineage specification in early development
Paper Link: https://www.nature.com/articles/s41422-023-00887-z
Editor:Li Han

